Five-Year Roadmap to Solving the Delivery Problem in the Brain and Spinal Cord: Unlocking CRISPR for C9 ALS and FTD
The CureC9 Program is a program within EverythingALS, a 501(c)(3) nonprofit. EverythingALS takes no overhead on CureC9 Program gifts. Every tax-deductible dollar flows through EverythingALS to the receiving academic institution, where we negotiate indirect costs below 10 percent — against a research-industry average closer to 60 percent. For a $1 million gift, this is the difference between roughly $400,000 and more than $900,000 reaching the bench.
Project Team — The Clelland Lab, UCSF

Principal Investigator: Claire Clelland, MD, PhD, MPhil
Title: John D. French Alzheimer’s Foundation Endowed Chair, UCSF Department of Neurology
Lab website: clellandlab.ucsf.edu
Bio page: clellandlab.ucsf.edu/people-0
Dr. Clelland is the John D. French Alzheimer’s Foundation Endowed Chair in the UCSF Department of Neurology. As a neurologist, she specializes in caring for patients with dementia and cognitive symptoms at the UCSF Memory & Aging Center. She is the recipient of national awards for neuroscience — the Alzheimer’s Association & NACC New Investigator Award, the American Neurologic Association’s Grass Foundation Award in Neuroscience, WashU’s Bold Pioneer Award.
The Clelland Lab at the UCSF Weill Institute for Neurosciences develops first-in-class therapies for neurodegenerative diseases. The lab is creating novel CRISPR gene therapies for C9orf72, the leading genetic cause of both ALS and frontotemporal dementia (FTD).
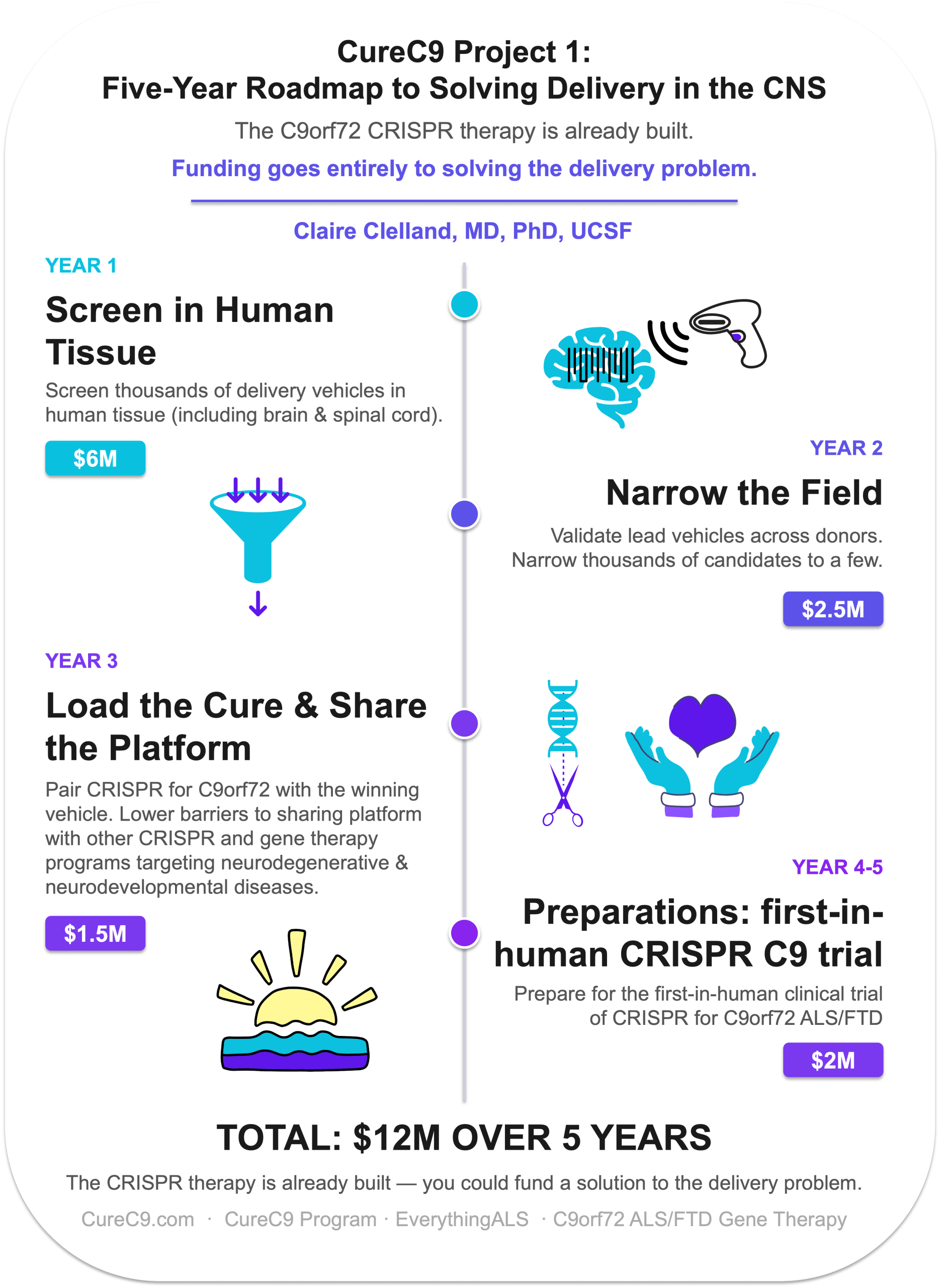
C9orf72 disease begins with a repeat expansion — an excess stretch of DNA that produces toxic RNAs and proteins, and drives both ALS and frontotemporal dementia (FTD). One promising approach is to cut out the excess DNA itself, using CRISPR as the tool. Dr. Clelland’s lab at UCSF has already done that part: they can remove the C9 mutation in cells from patients, and doing so eliminates the pathologic hallmarks of C9 disease. The remaining bottleneck — and the reason no CRISPR therapy for the brain exists yet — is delivery: getting CRISPR into enough cells in the brain and spinal cord to make a clinical difference. Over five years, with a $12 million investment, Dr. Clelland’s lab is ready to take on delivery. Solving it unlocks the first CRISPR gene therapy for C9 ALS/FTD, and the same delivery platform unlocks gene therapies for every other form of ALS and FTD, and for many other neurodegenerative and neurodevelopmental diseases that share this bottleneck.
Unlock gene therapy in the brain.

Simplified Explanation
A five-year plan to deliver the first CRISPR gene therapy for C9orf72 ALS and FTD. The CRISPR therapy itself is already built. Funding goes entirely to solving the delivery problem — identifying the optimal vehicle to shuttle CRISPR to the brain and spinal cord.
More Technical Details — Read the Clelland Lab’s full scientific write-up
Dr. Clelland, PhD, MD, MPhil, is the John D. French Alzheimer’s Foundation Endowed Chair in the UCSF Department of Neurology. As a neurologist, she specializes in caring for patients with dementia and cognitive symptoms at the UCSF Memory & Aging Center. Dr. Clelland is the recipient of national awards for neuroscience — The Alzheimer’s Association & NACC New Investigator Award, the American Neurologic Association’s Grass Foundation Award in Neuroscience, WashU’s Bold Pioneer Award.
Dr. Clelland’s lab at the UCSF Weill Institute for Neurosciences (clellandlab.ucsf.edu) develops first-in-class therapies for neurodegenerative diseases. The Clelland lab is creating novel CRISPR gene therapies for C9orf72, the leading genetic cause of both ALS and frontotemporal dementia (FTD). Using CRISPR, the Clelland lab has shown it is possible to remove the C9orf72 mutation in stem cells from patients and that this alleviates all of the pathologic hallmarks of C9-FTD/ALS in neurons derived from these cells. The therapy is currently being tested in mice. The team has also developed the necessary diagnostics, bioinformatic tools and off-target analysis to rapidly advance this therapy to clinical trial.
The biggest hurdle preventing the translation of CRISPR and other gene therapies from treating diseases of the brain and spinal cord, such as ALS and dementia, is the lack of effective delivery vehicles to shuttle these therapies to their target organ (called the “delivery problem”). The discovery of new delivery shuttles is hampered by the fact that animal models do not predict human outcomes, particularly for brain and spinal cord delivery. In the conventional path to the clinic, the first time that an engineered vehicle is evaluated in humans is when it is dosed in living patients. Dr. Clelland aims to upend this system by determining efficacy in humans early in the discovery process, before resource-intensive preclinical testing and first-in-human trials, by utilizing physiologically maintained cadavers. Testing hundreds to thousands of delivery vehicles in human tissue during the discovery phase of research will usher in a new age of human-based discovery research that will both create the next generation of delivery vehicles and build confidence in novel human therapies before use in living patients. This would revolutionize how human-specific discovery research is conducted. The Clelland lab, along with a team of innovative clinicians, neuroscientists, nurses, and ethicists, is ready to run large scale delivery studies to find suitable vehicles to shuttle CRISPR to the brain and spinal cord, enabling gene therapy for C9orf72 and other diseases body wide, potentially enabling therapies for millions of patients with fatal genetic diseases.

